Faecal volatilomics in exposome research
Novel Technologies used in HEDIMED
Volatile organic compounds (VOCs) are substances that readily evaporate in our ordinary living environment. This means that they have a high vapour pressure at room temperature and low boiling point, which facilitates their volatility and makes them responsible for odours and smells. VOCs are a diverse group of compounds derived mainly from biogenic sources, of which approximately 1.15 billion tonnes are released into the atmosphere each year. Terrestrial plants are responsible for 90% of this production. Anthropogenic emissions account for about 142 millions tonnes per year. 1,2.
In the HEDIMED project, our aim is to investigate a more modest biogenic source of VOCs. These are released from human faeces and have the potential to serve as biomarkers for a wide range of diseases and health conditions. Faecal volatilomics, a scientific research discipline that is dedicated to the in-depth investigation of the VOCs that emerge from faecal samples, is particularly promising because it is cost-effective and non-invasive – samples are generated daily and “just need to be collected”.
So-called ”electronic noses”
Electronic nose is a layman’s term for technologies designed to monitor VOCs. However, it’s important to be clear about what is meant by the term, as it can refer to different technologies in different contexts. In general, different electronic noses are sensitive to different odorants, with some exceptions. However, it’s important to realise that many of these technologies do not attempt to mimic the function of a real nose. Some compounds that have an odour to the human nose won’t give a signal to an electronic nose, and vice versa. This is the case with the technologies used in HEDIMED.
Differential Mobility Spectrometry (DMS)
Differential Mobility Spectrometry (DMS), is an instrument that utilises an electric mobility of ions for separation and identification of gas components.3 Ion mobility and its changes are influenced by, for example, the shape and size of the ion. Changing the electric field, which moves the ions between the high and low fields, creates a filter in which only some of the ions survive to reach the detector. The IonVision by Olfactomics, is the instrument that utilises DMS. See Picture 1. below.

DMS is typically used to measure gaseous samples. However, it is also possible to measure liquids and solids by measuring the gas space above the sample in a closed vessel. The latter method is used in HEDIMED, as shown in Picture 1. Usually, you want to find a substance in the sample that tells you something about the state of the sample. In this case, DMS can be used to investigate the presence of a defined substance or its concentration in the sample.

DMS has many advantages from a researcher’s perspective. DMS requires only a small amount of sample, typically 25-40 mg. There is no need for a preparation protocol such as adding solution, centrifugation etc. The analysis is also fast. One measurement takes only about 30 seconds per sample. In addition, the instrument is portable and easy to use.
In HEDIMED, the technology is used with the aims to create volatilomic ‘fingerprints’ in faecal samples of children at risk for type 1 diabetes and compare these with the ‘fingerprints’ of healthy controls. However, other methods are needed to identify which substances the fingerprints correspond to. For this, HEDIMED will also use complementary technologies, one of which will be introduced next.
Comprehensive two-dimensional gas chromatography-mass spectrometry
The other technique used to study faecal VOCs is comprehensive two-dimensional gas chromatography coupled with time-of-flight mass spectrometry (GC×GC/TOFMS). This can separate complex mixtures of volatile compounds and identify individual substances using a mass detector. It is very sensitive and has high resolution, making it a powerful tool for characterising the odour of faecal samples and potentially detecting compounds that may have previously gone unnoticed. It was selected for the HEDIMED project to complement other analytical approaches in metabolomics, such as liquid chromatography and nuclear magnetic resonance.
A great advantage of GC×GC/TOFMS is the variety of sample introduction options. The faecal odour (a rich mixture of VOCs) can be captured on a sorbent in the headspace above the sample and automatically evaporated from this sorbent into the chromatograph. (See the workflow in Picture 3.) Separation then takes place on two different chromatographic columns with different polarity and selectivity. After the substances in the carrier gas have passed through the first column and, in the case of complex mixtures, may not be completely separated, they are held at low temperature (condensed) for a short time (on the order of seconds) in the modulator and then evaporated again at high temperature and injected into the second column. This column, however, has a different polarity and selectivity and allows further separation of substances that have left the first column unseparated. From the second column, the compounds enter the TOF (time of flight) mass spectrometer.
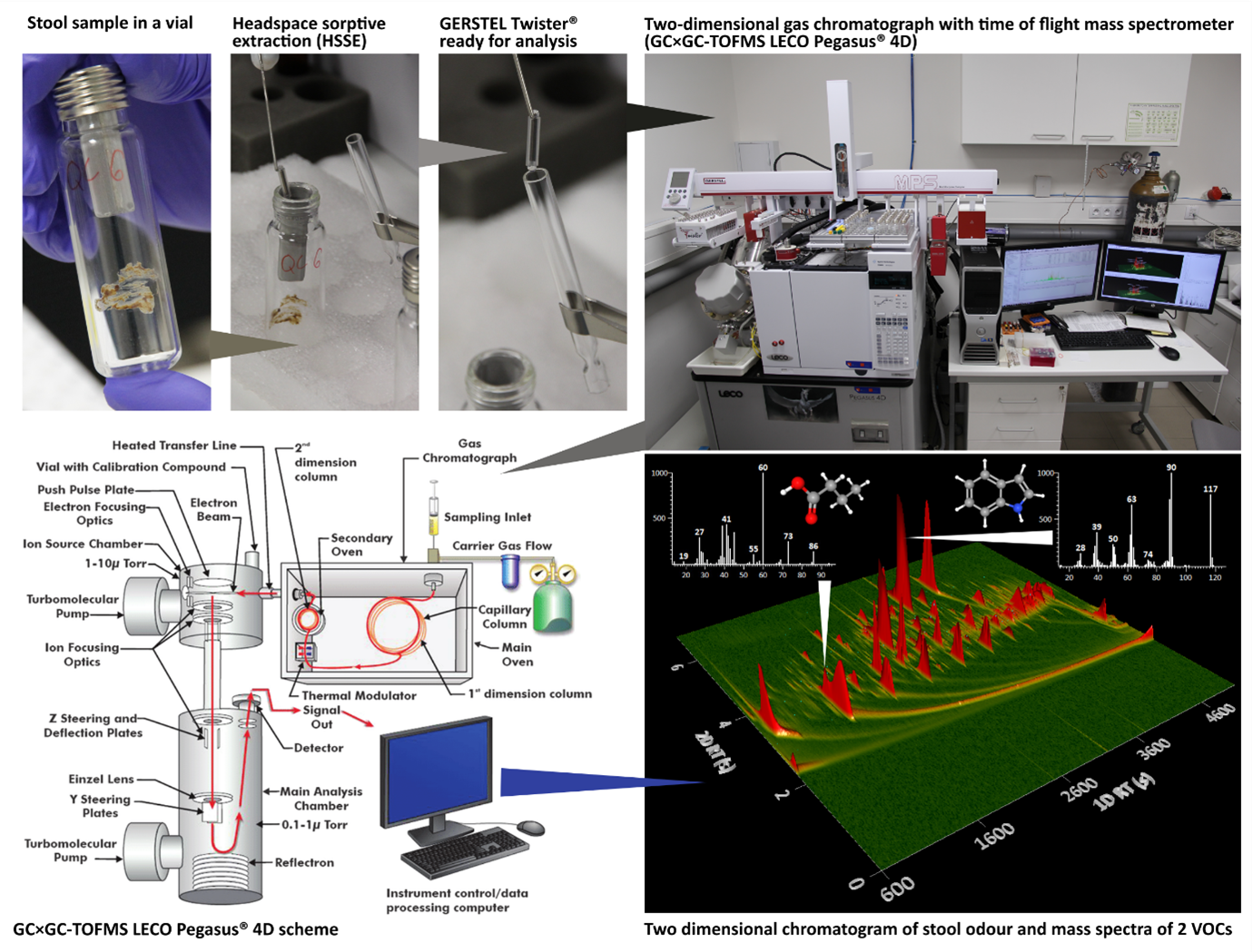
In the ion source they encounter a beam of electrons, become charged (ionised) and, due to the excess energy in the chemical bonds, begin to fragment into specific ions according to the nature of the molecule. The group of ions produced by the fragmentation of the molecule is then propelled by an electrical impulse into the evacuated flight tube and flies towards the detector, where they arrive in order from lightest to heaviest. The masses of the individual ions produced by the fragmentation of the molecule are calculated from the time of flight and the length of the path, and their quantity is recorded. This gives a fragmentation mass spectrum for each substance, which can be thought of as a fingerprint of the molecule (see Picture 3.). The visual result of the analysis is a two-dimensional chromatogram resembling a mountain range with peaks. Each of these peaks ideally represents a single compound for which we have measured a mass spectrum, and the higher the peak, the more of that compound there was.
Its sensitivity and high resolution make GC×GC/TOFMS an excellent tool for volatilomics. The amount of sample required is very small, around 10 mg, and the preparation protocol is simple. However, GC×GC/TOFMS is considered a traditional research method and requires laboratory facilities and expertise to operate.
To get more information about the technologies, don’t hesitate to contact the experts!
GC×GC/TOFMS: Jan Rezek, the Institute of Experimental Botany of the Czech Academy of Sciences, email: rezek@ueb.cas.cz
DMS: Meri Mäkelä, Olfactomics, email: meri.makela@olfactomics.fi
COntributions
Planning and drafting: Minna Turppa and Eliška Jeníčkova Introduction: Jutta Laiho, Jan Rezek, Minna Turppa and Eliška Jeníčková DMS section: Meri Mäkelä, Minna Turppa, Eliška Jeníčková and Jutta Laiho GC×GC/TOFMS section: Jan Rezek and Eliška Jeníčková
Pictures: 1 Meri Mäkelä and Eliška Jeníčková, 2 Meri Mäkelä, 3 Jan Rezek
References:
- Goldstein A, Galbally I (2007) Known and Unexplored Organic Constituents in the Earth’s Atmosphere. Environ Sci Technol 41:1515–1521. https://doi.org/10.1021/es072476p
- Sindelarova K, Granier C, Bouarar I, Guenther A, Tilmes S, Stavrakou T, Müller JF, Kuhn U, Stefani P, Knorr W (2014) Global data set of biogenic VOC emissions calculated by the MEGAN model over the last 30 years. Atmos Chem Phys 14:9317–9341. https://doi.org/10.5194/acp-14-9317-2014
- Olfactomics. 2024. DMS technology. Available: https://olfactomics.fi/technology/






